ISSUE1698
- Mark Abramowicz, M.D., President has disclosed no relevant financial relationships.
- Jean-Marie Pflomm, Pharm.D., Editor in Chief has disclosed no relevant financial relationships.
- Amy Faucard, MLS, Associate Editor has disclosed no relevant financial relationships.
- Review the efficacy and safety of Airsupra, a combination of the inhaled short-acting beta2-agonist albuterol and the inhaled corticosteroid budesonide, for as-needed treatment or prevention of bronchoconstriction.
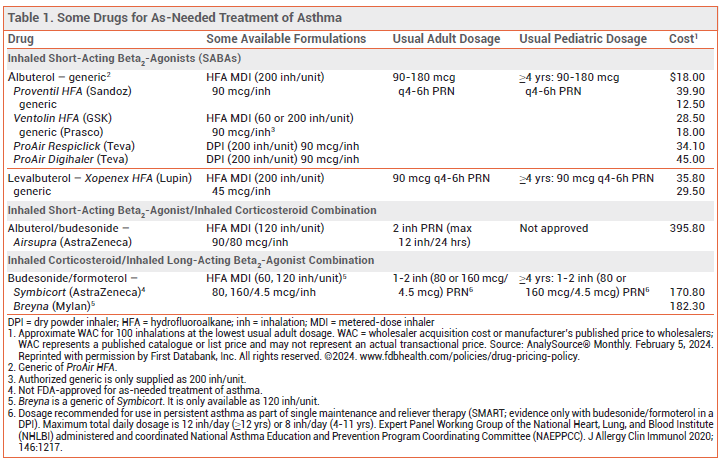
- Description: A fixed-dose combination of the inhaled short-acting beta2-agonist albuterol and the inhaled corticosteroid budesonide.
- Indication: For use as needed for treatment or prevention of bronchoconstriction and to reduce the risk of exacerbations in adults with asthma.
- Efficacy: Reduced the risk of a severe exacerbation compared to albuterol alone in adults with moderate to severe asthma.
- Adverse Effects: Most common are headache, dysphonia, oral candidiasis, and cough.
- Dosage: 2 inhalations of albuterol 90 mcg/budesonide 80 mcg as needed (max 12 inhalations/24 hours).
- Cost: 100 inhalations (50 doses) cost about $396.
- Conclusion: Treatment guidelines now recommend adding an inhaled corticosteroid to a beta2-agonist for as-needed use as a reliever therapy in adolescents and adults with asthma. The combination of the ICS budesonide and the long-acting beta2-agonist formoterol (Symbicort, Breyna) is an effective and less expensive alternative to Airsupra for such use.
Outline
Table |
The FDA has approved Airsupra (AstraZeneca), a metered-dose inhaler containing the short-acting beta2-agonist (SABA) albuterol and the inhaled corticosteroid (ICS) budesonide, for use as needed for treatment or prevention of bronchoconstriction and to reduce the risk of exacerbations in patients ≥18 years old with asthma. Airsupra is the first combination of a SABA and an ICS to become available in the US. It is not approved for use as maintenance therapy.

STANDARD TREATMENT ― The inhaled SABAs albuterol and levalbuterol are used as needed for rapid relief of asthma symptoms and for prevention of exercise-induced bronchoconstriction (EIB). Their bronchodilatory effect begins within 5 minutes after inhalation, peaks within 15-60 minutes, and lasts for 4-6 hours. They do not decrease airway inflammation.1
Treatment guidelines now recommend that most adolescents and adults with asthma use reliever therapies that include an anti-inflammatory component because addition of an ICS has been shown to reduce the risk of severe exacerbations compared to albuterol alone.2,3 Options for as-needed use include using a low-dose ICS and a SABA together (in a single inhaler [Airsupra] if possible) or using a combination of a low-dose ICS and the long-acting beta2-agonist (LABA) formoterol in a single inhaler (e.g., budesonide/formoterol [Symbicort, Breyna]; off-label). The onset of action of formoterol is similar to that of albuterol. In patients who require daily maintenance therapy, a low- to medium-dose ICS/formoterol combination can be used as single maintenance and reliever therapy (SMART). As-needed use of an ICS and a SABA in a single inhaler in addition to ICS/LABA maintenance therapy is an alternative, but it requires use of two separate inhalers.3,4
CLINICAL STUDIES ― Approval of albuterol/budesonide for adults with asthma was based on the results of two double-blind trials (MANDALA and DENALI). Small numbers of patients <18 years old were included in the trials; more data are needed to determine the efficacy of the combination in children and adolescents.
The MANDALA trial included 2940 adults with moderate to severe asthma. Patients were randomized to receive albuterol/budesonide or albuterol alone as needed (in addition to their daily maintenance therapy) for at least 24 weeks.
A first severe asthma exacerbation, the primary endpoint, occurred in 20% of patients who used albuterol 180 mcg/budesonide 160 mcg (the FDA-approved dose) and in 26% of those who used albuterol 180 mcg alone, a statistically significant difference. Use of the combination was also associated with a significantly lower annualized severe exacerbation rate (0.46 vs 0.60) and annualized systemic corticosteroid dose (87 mg vs 127 mg).5,6
In the DENALI trial in 964 adults with mild to moderate asthma, the median time to onset of bronchodilation (≥15% increase in post-inhalation FEV1) and mean duration of bronchodilation were similar with albuterol 180 mcg/budesonide 160 mcg and albuterol 180 mcg alone (7.5 and 186.9 minutes vs 10.0 and 167.9 minutes).7
In a double-blind, single-dose crossover study (TYREE) in 60 patients ≥12 years old with a history of asthma and EIB, use of albuterol 180 mcg/budesonide 160 mcg was more effective than placebo in preventing EIB.8
ADVERSE EFFECTS — The most common adverse effects of albuterol/budesonide in clinical trials were headache (4-5%), dysphonia (2%), oral candidiasis (1.3-1.5%), and cough (1%).
Inhaled SABAs can cause throat irritation, tremor, tachycardia, QT-interval prolongation, hyperglycemia, hypokalemia, and hypomagnesemia, especially when used in high doses. ICS use has been associated with nasopharyngitis, nasal congestion, pharyngitis, rhinitis, respiratory tract infection, gastroenteritis, and otitis media. Cataracts, glaucoma, and decreased bone mineral density have been observed with long-term use.
DRUG INTERACTIONS ― Budesonide is metabolized by CYP3A4; coadministration of a CYP3A4 inhibitor could increase its serum concentrations.9 Use of albuterol with or within 2 weeks of a monoamine oxidase inhibitor (MAOI) or a tricyclic antidepressant could increase the risk of cardiovascular adverse effects. Coadministration of a beta blocker can reduce the clinical efficacy of albuterol. Concomitant use of albuterol, especially in high doses, may worsen the hypokalemia and ECG changes caused by non-potassium-sparing diuretics.
PREGNANCY AND LACTATION — Albuterol and budesonide are considered safe for use during pregnancy. In epidemiological studies and case reports, use of the drugs in pregnant women has not been associated with an increased risk of major birth defects or miscarriage. Poorly controlled asthma symptoms and acute exacerbations increase the risk of pregnancy complications.
No data are available on the effects of albuterol/budesonide on the breastfed infant or milk production. Levels of albuterol following inhalation are likely to be low in human breast milk. In one study, small amounts of budesonide were detected in human milk following maternal use of inhaled budesonide, but no adverse events were observed in the breastfed infants.
DOSAGE AND ADMINISTRATION ― Airsupra is supplied as a pressurized metered-dose inhaler containing 120 inhalations. Each inhalation delivers 90 mcg of albuterol and 80 mcg of budesonide. The recommended dosage is 2 inhalations as needed for asthma symptoms (maximum 12 inhalations/24 hours). The inhaler must be primed before first use by releasing 4 sprays. It must be reprimed by releasing 2 sprays when it has not been used for >7 days, when it is dropped, or after cleaning.
CONCLUSION ― The fixed-dose combination inhaler containing albuterol and budesonide (Airsupra) is the first combination of a short-acting beta2-agonist (SABA) and an inhaled corticosteroid (ICS) to be approved in the US. As-needed use of the combination in addition to daily maintenance therapy significantly reduced the risk of a severe exacerbation compared to albuterol alone in adults with moderate to severe asthma; whether it is more effective than albuterol alone in children or adolescents with asthma remains to be established. Albuterol/budesonide has not been compared to budesonide/formoterol (Symbicort, Breyna), which has been shown to be effective (off-label) for this indication and costs less.
- Drugs for asthma. Med Lett Drugs Ther 2020; 62:193.
- I Crossingham et al. Combination fixed-dose beta agonist and steroid inhaler as required for adults or children with mild asthma. Cochrane Database Syst Rev 2021; 5:CD013518. doi:10.1002/14651858.cd013518.pub2
- Global Initiative for Asthma. Global strategy for asthma management and prevention, 2023. Updated July 2023. Available at: www.ginasthma.org. Accessed February 29, 2024.
- Expert Panel Working Group of the National Heart, Lung, and Blood Institute (NHLBI) administered and coordinated National Asthma Education and Prevention Program Coordinating Committee (NAEPPCC). 2020 focused updates to the asthma management guidelines: a report from the National Asthma Education and Prevention Program Coordinating Committee Expert Panel Working Group. J Allergy Clin Immunol 2020; 146:1217. doi:10.1016/j.jaci.2020.10.003
- A Papi et al. Albuterol-budesonide fixed-dose combination rescue inhaler for asthma. N Engl J Med 2022; 386:2071. doi:10.1056/nejmoa2203163
- FDA Briefing Document. Budesonide/albuterol sulfate metered dose inhaler. Pulmonary-Allergy Drugs Advisory Committee meeting 11/08/22. Available at: https://bit.ly/3ThFM6Y. Accessed: February 29, 2024.
- BE Chipps et al. Albuterol-budesonide pressurized metered dose inhaler in patients with mild-to-moderate asthma: results of the DENALI double-blind randomized controlled trial. Chest 2023; 164:585. doi:10.1016/j.chest.2023.03.035
- C LaForce et al. Albuterol/budesonide for the treatment of exercise-induced bronchoconstriction in patients with asthma: the TYREE study. Ann Allergy Asthma Immunol 2022; 128:169. doi:10.1016/j.anai.2021.10.020
- Inhibitors and inducers of CYP enzymes, P-glycoprotein, and other transporters. Med Lett Drugs Ther 2023 January 25 (epub). Available at: www.medicalletter.org/downloads/CYP_PGP_Tables.pdf.